Replacing Metals with 3D Printed Polymers

Introduction
Replacing metal with polymers is not only possible but becoming increasingly more prevalent. The development of high-performance polymers and composites has made this possible with materials that have similar mechanical properties to aluminum at a fraction of the weight.
In addition, the ability to additively manufacture parts from these polymers, with high-temperature 3D printers like the AON M2+, has driven further adoption by reducing the complexity, lead times, and cost constraints of traditional manufacturing methods.

What differentiates metals and polymers?
Before we address which polymers are suitable for metal substitution, we need to set the right expectations. Some companies may inflate the capabilities of high-performance polymers and fail to consider that metals and polymers are fundamentally different. Here’s how they differ.
Polymers
3D printable polymers are thermoplastics, made up of polymer chains that are chemically separate from each other (i.e., no molecular bonding between chains). These chains are physically entangled which provides mechanical integrity to the overall material. If these chains are organized into a crystal structure, the chains are harder to pull apart. Therefore, semi-crystalline polymers are better in strength and stiffness compared to amorphous polymers.
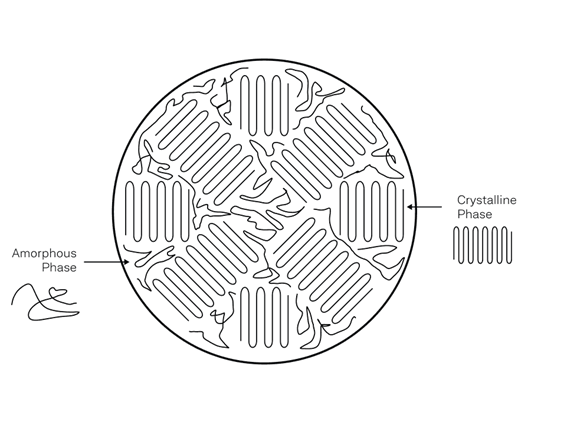
Metals
If you were to put a common metal under the microscope, you’d see a structure similar to image A below. Metals are made up of small grains, where each grain consists of a crystal lattice (image B). Often, these grains differ from each other by the size and orientation of the crystal structure. The crystals meet at grain boundaries, where strong metallic bonding is the main force of attraction between atoms.

Polymers vs Metals: Conclusion
In conclusion, we can say that:
- semi-crystalline polymers are partially made of crystals,
- while metals contain many crystal structures (grains) that differ in orientation.
Since thermoplastic polymer chains are chemically separate, the attraction force between chains is weaker than the metallic bonding between crystals in metals. Therefore, metals are more resistant to elastic deformation (stiffer) and generally more temperature resistant. It’s the reason why metals have been the default choice for mechanical and thermal applications.
However, as more advanced thermoplastics are engineered, the effective strength begins to match and, in some cases, surpasses that of metals.
Two Suitable Metal Replacement Materials: PEEK and ULTEM™
While PEEK and ULTEM™ 9085 do not represent every high-performance polymer capable of metal replacement, they are two of the most popular.
In the graphs below, along with Carbon Fiber PEEK, we compare these materials to Aluminum 6061, a common alloy used in the construction of aircraft structures, automotive parts, marine structures, and consumer products.
These are several industries where metal replacement can provide significant weight savings while maintaining the required mechanical thermal properties, plus added chemical resistive properties.

Specific strength is used to evaluate the tensile strength of a material compared to its density. The higher specific strength of thermoplastics allows for lighter-weight parts while maintaining the same strength properties of metals.

Via the above graphs, you can see that PEEK and ULTEM™ offer significant weight reduction– between 50-60% lighter than aluminum– while maintaining high strength and high heat resistance.
In many industries, such as transportation, replacing metals means greater fuel savings, lower carbon emissions, or increased payload capabilities. In addition, these materials possess high chemical resistance, high impact resistance, are UL94-V0 flammability rated, and meet FAR 25.853 flame, smoke, and toxicity (FST) requirements, all necessary in aerospace and naval applications.
How to 3D Print PEEK and ULTEM™
Simply 3D printing high-performance materials does not guarantee final parts will possess the same mechanical, thermal, and chemical properties of the parent polymer.
To print these materials, a 3D printer needs a thermally optimized, precision-controlled, high-temperature build chamber (min. 132°C) and 500°C extruders, and configurable print surfaces are highly recommended. Printing at temperatures colder than 132°C can significantly reduce part strength, chemical resistance, and require further annealing in which warping is unavoidable.
AON3D AON M2+ overview
The AON M2+ is a 3D printer produced by AON3D, a manufacturer based in Canada.
It uses the Fused Filament Fabrication technology to produce elastomers and thermoplastics parts using filament feedstock.
It offers a build volume of 450 × 450 × 565 mm.
The AON M2+ is a large, high-temperature industrial 3D printer optimized to make 3D printing high-performance materials, like PEEK and ULTEM™ 9085, easy and accessible. Learn more about 3D printing PEEK, PEKK, and ULTEM™ in AON3D’s webinar.
 English
English  Français
Français


